Question
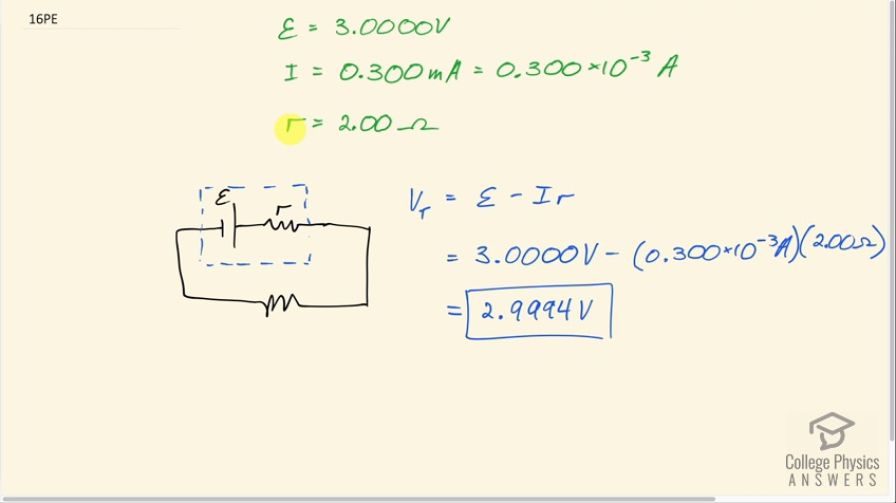
What is the output voltage of a 3.0000-V lithium cell in a digital wristwatch that draws 0.300 mA, if the cell’s internal resistance is ?
Final Answer
Solution video
OpenStax College Physics for AP® Courses, Chapter 21, Problem 16 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. A lithium-ion battery in a wrist watch has an emf of 3.0000 volts and an internal resistance of 2.00 ohms and we are told that the current being drawn from this battery by the watch circuit is 0.300 milliamps, which I write as 0.300 times 10 to the minus 3 amps. So the terminal voltage is the question: what is this voltage between the terminals of the battery? And that's going to equal the emf minus the voltage drop across the internal resistance, which is the current multiplied by r. So that's 3.0000 volts minus 0.300 times 10 to the minus 3 amps times 2.00 ohms and this is 2.9994 volts.
