Question
Bird bones have air pockets in them to reduce their weight—this also gives them an average density significantly less than that of the bones of other animals. Suppose an ornithologist weighs a bird bone in air and in water and finds its mass is and its apparent mass when submerged
is (the bone is watertight). (a) What mass of water is displaced? (b) What is the volume of the bone? (c) What is its
average density?
Final Answer
Solution video
OpenStax College Physics for AP® Courses, Chapter 11, Problem 40 (Problems & Exercises)
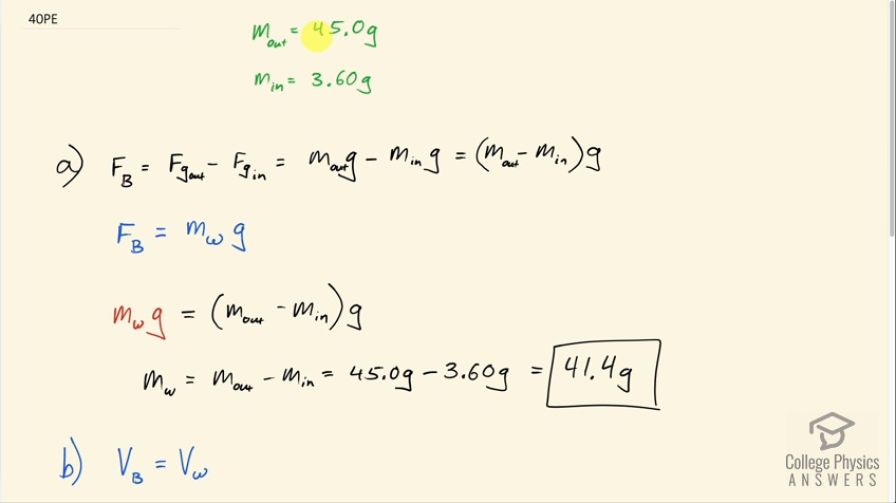
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. The mass of some bird bones is measured in air outside of some water and the mass is 45.0 grams and the apparent mass in the water is 3.60 grams and the first question is what is the mass of water displaced? So the buoyant force is this difference between the weight of the bones out of the water versus in the water and so that is the mass out of the water times g minus the mass in the water times g and then g can be factored out and the buoyant force also equals the weight of fluid displaced— that's Archimedes principle— and so that's the mass of water displaced times g so we can equate this with this then... we can replace F B with m wg and say it's equal to the difference between the mass of the bones out and in the water times g. The g's cancel and so the mass of water displaced is the difference in these mass measurements for the bones and that is 41.4 grams. The next question is what is the volume of the bones? Well the volume of the bones is gonna equal the volume of water displaced and we know that density of water is the mass of the water displaced— which we have found— divided by the volume of water and we can solve for V w by multiplying both sides by V w divided by ρ w. And so the volume of water displaced then is the mass of the water displaced divided by density of water and we can replace V w with that here. So the volume of the bone then is 41.4 grams of water displaced divided by 1 gram per milliliter—density of water— and that is 41.4 milliliters then. Part (c) asks what is the average density of the bone? It's going to be the mass of the bone divided by its volume so that's 45.0 grams divided by 41.4 milliliters and that is 1.09 grams per milliliter.
