Question
A 76.0-kg person suffering from hypothermia comes indoors and shivers vigorously. How long does it take the heat transfer to increase the person’s body temperature by if all other forms of heat transfer are balanced?
Final Answer
Solution video
OpenStax College Physics for AP® Courses, Chapter 14, Problem 71 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
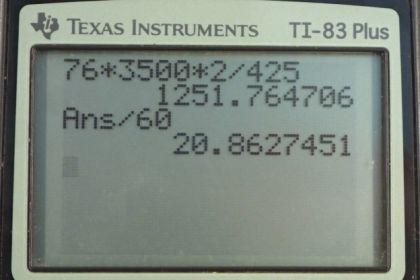
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. So, a person comes indoors and they're cold and they're going to be shivering and they're going to increase their body temperature by 2 Celsius degrees. The question is how long will it take them to do that, assuming their mass is 76 kilograms? Now the rate at which energy is being produced by shivering is 725 watts and we're told that there's no heat transfer due to any other mechanism, be it radiation or conduction and so on. So, I had to look up this number in the textbook in Table 7.5. It says that shivering creates an energy output of 425 watts. So, power is heat or energy transferred divided by time. And, we'll solve for T by multiplying both sides by T over P. So, time is going to be the energy that is consumed to heat them up divided by the rate at which it is generated by the shivering. So, that's mass times specific heat of the body times the change in temperature divided by P, which is 76 kilograms times 3500 Joules per kilogram per Celsius degree specific heat of the body times 2 Celsius degrees change in temperature divided by 425 watts of heat generated. And, that is this many seconds which we convert into minutes by multiplying by one minute for every 60 seconds, and this works out to 20.9 minutes for their shivering to increase their body temperature by 2 Celsius degrees.