Question
An x ray tube has an applied voltage of 100 kV. (a) What is the most energetic x-ray photon it can produce? Express your answer in electron volts and joules. (b) Find the wavelength of such an X–ray.
Final Answer
Solution video
OpenStax College Physics, Chapter 30, Problem 27 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
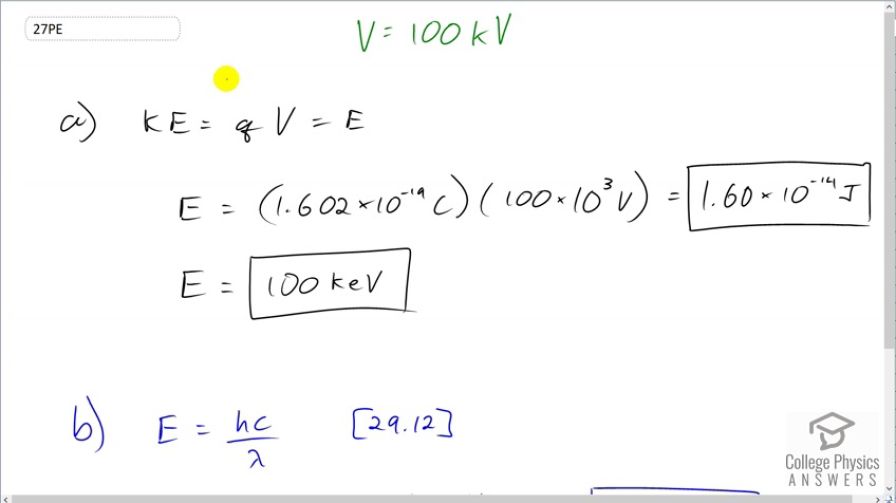
This is College Physics Answers with Shaun Dychko. An x-ray tube a voltage of 100 kilovolts and the maximum energy photon that it can produce is equal to the kinetic energy of the electron that's accelerated through the tube. and when that electron hits the anode at the other end of the tube, it can produce x-rays of a variety of different wavelengths but the maximum would be one that has an energy that is equal to the kinetic energy of the electron. So let's calculate what that kinetic energy of the electron is it's q times V; that's the elementary charge times the voltage of the x-ray tube and that's going to be the energy of the photon and we'll multiply elementary charge by 100 times 10 to the 3 volts giving 1.60 times 10 to the minus 14 joules or as in units of electron volts, we can call this 100 kiloelectron volts because well, you could multiply this by 1 electron volt for every 1.602 times 10 to the minus 19 joules but then you would be canceling with this leaving you with this but with units of electron volts and it's times 10 to the 3 so it's kiloelectron volts. So the maximum energy photon produced by an x-ray tube is always the x-ray tube voltage written with units of electron volts instead of volts. The wavelength of this maximum energy photon is given by this formula [29.12] which is Planck's constant times speed of light divided by λ. We'll multiply both sides by λ over E and we get a λ is hc over E. So that's 1240 electron volt nanometers and we'll divide that by 100 times 10 to the 3 electron volts giving us units of nanometers that is 0.0124 nanometers.
