Question
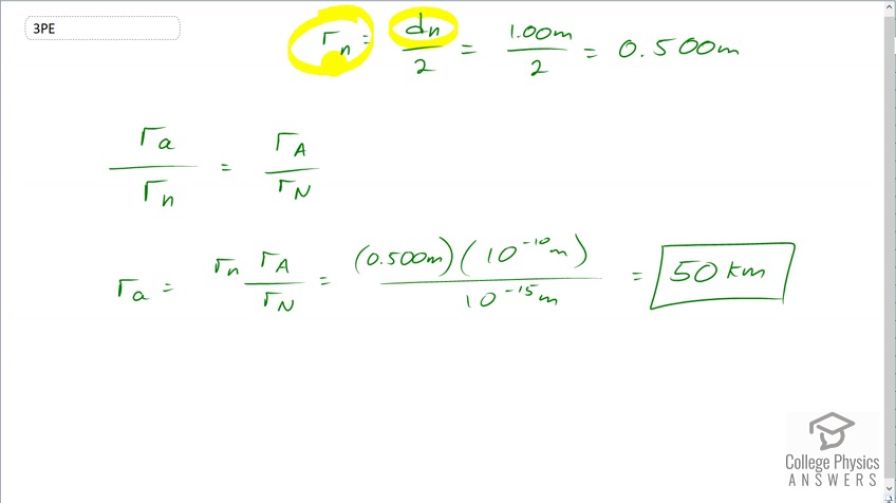
If someone wanted to build a scale model of the atom with a nucleus 1.00 m in diameter, how far away would the nearest electron need to be?
Final Answer
Solution video
OpenStax College Physics, Chapter 30, Problem 3 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. Given a scale model with a nucleus having a diamter of 1 meter, we can figure out the radius of this hypothetical nucleus which would be half of that which is 0.5 meters. Now we know what the ratio is of the atom to a nucleus and an atom is typically about 10 to the minus 10 meters wide and the nucelus is 10 to the minus 15 meters wide. And so we are gonna have the same ratio in our scale model so the radius of our scale atom divided by the radius of our scale nucleus has to have the same ratio as for real atoms. And so we'll multiply both sides by the radius of our scale nucleus and we solve for the radius of our scale atom. So that's r little n times r capital A over r capital N; that's 0.500 meters times 10 to the minus 10 meters over 10 to the minus 15 meters which is 50 kilometers. So having a nucleus of 1 meter to scale would need an atom of radius 50 kilometers.
