Question
In 1986, a gargantuan iceberg broke away from the Ross Ice Shelf in Antarctica. It was approximately a rectangle 160 km long, 40.0 km wide, and 250 m thick.
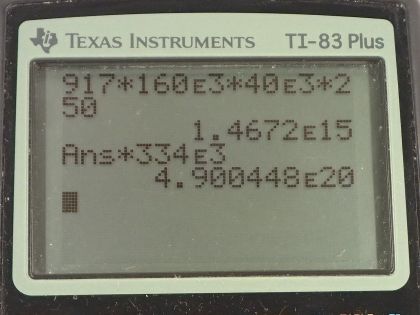
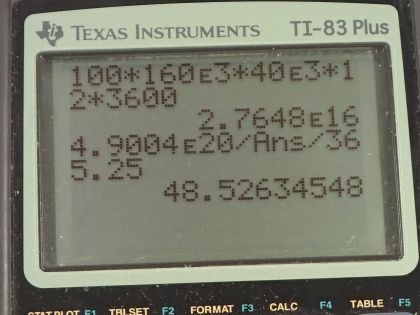
(a) What is the mass of this iceberg, given that the density of ice is ? (b) How much heat transfer (in joules) is needed to melt it? (c) How many years would it take sunlight alone to melt ice this thick, if the ice absorbs an average of , 12.00 h per day?
Final Answer
Solution video
OpenStax College Physics, Chapter 14, Problem 18 (Problems & Exercises)

vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. A gargantuan iceberg broke off from the Ross Ice Shelf in Antarctica in 1986 and it had a length of 160 kilometers, a width of 40.0 kilometers and a thickness or depth of 250 meters and I have converted these kilometers into meters by multiplying by 10 to the 3 since that's what the prefix 'kilo' means. So in part (a), it asks us what the mass of this iceberg is? So density is what we are given and it's mass divided by volume and we can solve for mass by multiplying both sides by V and so the mass is density times volume. The volume is length times width times depth and we multiply that by ρ so that's 917 kilograms per cubic meter—density— times the length times the width times the depth and that's 1.47 times 10 to the 15 kilograms. Part (b) asks how much heat energy will it take to melt it? So that's gonna be its mass times the latent heat of fusion for water which we look up over here in table [14.2] and we see that water has a latent heat of fusion of 334 kilojoules per kilogram and so the 'kilo' means times 10 to the 3 so we have 334 times 10 to the 3 joules per kilogram here multiplied by the mass we found in part (a) and that's 4.90 times 10 to the 20 joules. In part (c), it tells us what the solar flux is— the power per square meter of the sunlight— and how many years would it take the energy from the sunlight to melt this iceberg. So we have to figure out how many joules are being absorbed per day so that's 100 watts per square meter and we multiply that by the area of the iceberg which is its length times its width and then multiply by 12 hours per day— we assume that the Sun is shining for 12 hours everyday— and this gives us the number of watts per day but then we have to multiply by 3600 seconds per hour in order to turn this into joules per day because this watts is actually joules per second and so these seconds are gonna cancel with these seconds and we get them by turning this hour into seconds and we are left with this day on the bottom though because this is the number of joules absorbed per day. Okay! So that's 2.7648 times 10 to the 16 joules absorbed everyday and so the total time to melt then will be the number of joules that are needed— that we calculated in part (b)— multiplied by 1 day for every 2.7648 times 10 to the 16 joules. I decided to multiply by the reciprocal of this number; you could also divide by this number and it would be the same. And then that will give us units of days for our time so we can write that into years by multiplying by 1 year for every 365.25 days and that's 48.5 years for this iceberg to melt assuming the only place it got heat energy from was the Sun which in the real world is not the case, it's also gonna absorb a lot of heat energy from the surrounding water so it will take less time than this in reality.