Question
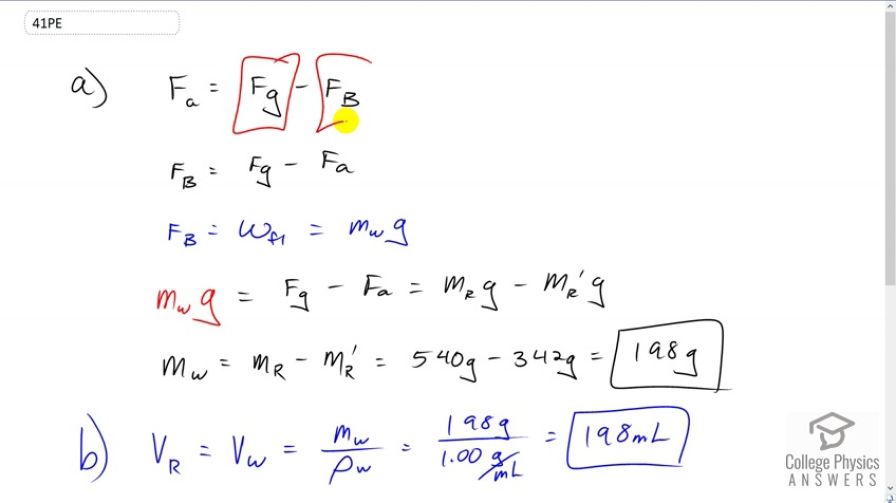
A rock with a mass of 540 g in air is found to have an apparent mass of 342 g when submerged in water. (a) What mass of water is displaced? (b) What is the volume of the rock? (c) What is its average density? Is this consistent with the value for granite?
Final Answer
- 198 g
- 198 mL
- 2.73 g/mL
Yes, this is granite.
Solution video
OpenStax College Physics, Chapter 11, Problem 41 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. The apparent weight when this rock is in water equals its weight when it's not in water minus the buoyant force the water provides. We can solve for the buoyant force by adding F b to both sides, then also subtracting F a from both sides. So we get F b on the left equals force of gravity minus the apparent weight. The buoyant force also is the weight of the fluid displaced, that's Archimedes principle, so that will be the mass of the fluid displaced, multiplied by g. We're going to be solving for this m subscript w, this mass of water displaced in this question part A. So we'll substitute this in place of the buoyant force and so we have m w g equals F g minus F a. F g is the mass of the rock times g, minus the apparent mass of the rock times g and the g's cancel on both sides. We're left with m w is the difference between the rock's mass out of the water and in the water. So that's 540 grams minus 342 grams which is 198 grams will be the mass of water displaced. The volume of the rock is going to be the volume of water displaced because the rock is submerged entirely. So the volume of the water is going to be its mass divided by its density. So that's 198 grams divided by one gram per milliliter which is 198 milliliters. Then part C is what is its average density? Well we have to take the mass of the rock when it's out of the water, so its real mass so to speak, divided by its volume. So that's 540 grams divided by 198 milliliters and that works out to 2.73 grams per milliliter. When you look at the table of densities in the textbook here, yes, this density is granite. So this rock is made out of granite.
