Question
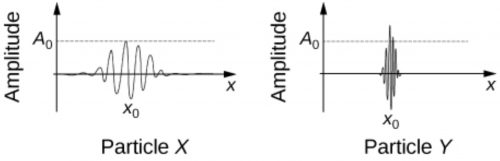
In Figure 30.66, explain qualitatively the difference in the wave functions of particle X and particle Y. Which particle is more likely to be found at a larger distance from the coordinate x0 and why? Which particle is more likely be found exactly at x0 and why?
Final Answer
Please see the solution video.
Solution video
OpenStax College Physics for AP® Courses, Chapter 30, Problem 14 (Test Prep for AP® Courses)
vote with a rating of
votes with an average rating of
.
Video Transcript
This is College Physics Answers with Shaun Dychko. This picture shows a wave function for particle X and a different wave function for particle Y and the question is which particle is more likely to be found at a larger distance from the coordinate x naught? And the answer is particle X will be found a larger distance away because its wave function describes the probability of finding particle at each of these positions. Now strictly speaking, it's the square of— the absolute value of the wave function— that is the probability so the probability would be something like this and this probability is quite spread out so at a far distance from x naught like all the way over here you know, this might be... let's call this Δx— this is a large spread— versus particle Y will have its probability distribution like this from the mid-point x naught to the end of this bell curve— this is a smaller interval in x— so we'll call this Δx Y and this is Δx X and we can say that Δx X is bigger than Δx Y and this is our explanation for how we know that particle X is more likely to be some far distance away from the position x naught. Which particle is more likely to be found exactly at x naught? Okay! Well, the answer to that question is particle Y and because this wave function is horizontally compressed and so this probability of finding it is narrowly constrained close to this position x naught.